Can Utility-Scale Batteries Rescue Intermittent Renewables? (Improvement, market shakeout, but no ‘silver bullet’)
By Robert Peltier -- March 10, 2010All interconnected transmission and distribution (T&D) grids have one thing in common. Their operators must continually dispatch generators to keep the network’s supply and demand in balance at all times and to maintain its voltage and frequency within very tight tolerances.
The “simultaneity problem” is not shared by oil or natural gas or coal. It is a tough reality for electricity that Thomas Edison and countless inventors since him have tried to solve via affordable battery storage.
So where are we today in terms of cost per kWh to use batteries to store power and, in the case of intermittent technologies, firm power? For utility scale battery systems, expect to pay between $1,000/kW and $4,000/kW, according to the Electricity Storage Association. The DOE’s optimistic assessment estimates those costs will drop to around $500/kW by 2012.
Such adds at least a half cent per kWh to the cost of electricity.
Latest Technologies
There are about a dozen technologies vying for a piece of the utility-scale energy storage market, especially advanced battery technologies such as lithium ion and sodium sulfur batteries, pumped hydro, and compressed air energy storage. In this post, we’ll review the state-of-the-art of battery technology, a few interesting projects, and get a glimpse of the next generation of utility-scale batteries.
You should also note the few U.S. projects over the past few years and the large number number of battery technology companies chasing those projects. Several companies have since left the battery market or redefined their products. Little data on installed costs is available but included when available. Expect a major market shake-out over the next year or two.
The ongoing dissolution of the traditional electricity sector structure also seems to call for increased reliance on big batteries wherever feasible. One consequence of deregulation is that, in many states, generation and T&D are no longer planned in an integrated fashion by one entity—the local utility. Energy storage in general, and batteries in particular, can help stabilize the intermittent nature of nondispatchable renewable energy sources, for load leveling and peak shaving, substation standby power, or as fast acting reserves for system regulation control (ancillary services). Storage also has a critical role to play in securing the nation’s energy infrastructure, much as the Strategic Petroleum Reserve does for oil, and bulk gas storage does for balancing seasonal natural gas demand and supply.
Many Battery Technologies
New and rechargeable sodium-sulfur, vanadium redox, and zinc-bromide batteries—as well as nickel-cadmium batteries, which went commercial decades ago—have already demonstrated their ability to act as generators as large as 10 MW but the capacity must grow substantially to meet the growing need for reliably energy storage.
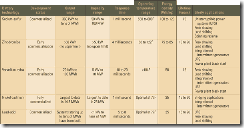
Table 1 summarizes many of the available energy storage options and the status of and future prospects for several battery technologies that either currently offer bulk energy storage capability or promise to do so in the near future. The following discussions and case studies explain how the new bulk-storage batteries work and how pioneers are using them to solve a number of common T&D problems.
1. A comparison of today’s bulk electricity storage technologies. Source: Platts
Sodium-sulfur: A Hot New Battery
Ford Motor Co. is credited with launching the development of the sodium-sulfur (NaS) battery in the 1960s for vehicular applications. However, today’s next-generation NaS batteries are the end result of more than a decade of R&D by NGK Insulators, Ltd. and Tokyo Electric Power Co. (Tepco). Tepco had identified the technology as the best candidate for replacing central pumped-hydro energy storage in Japan. NGK, which supplied the expertise in ceramics that was crucial to the development of the technology, is today the only supplier of NaS batteries for bulk storage applications.
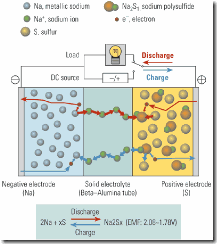
Sodium-sulfur batteries use molten sulfur as the positive electrode and molten sodium as the negative electrode. These active materials are separated by a solid, ceramic electrolyte that conducts sodium ions (Figure 1).
1. Sodium-sulfur (NaS) battery operation. A solid ceramic electrolyte separates the liquid sulfur positive electrode from the liquid sodium negative electrode. Source: NGK Insulators Ltd.
During discharge, positive sodium ions flow through the electrolyte to combine with the sulfur, forming sodium polysulfides. Electrons flow through the external circuit of the battery to create a potential difference. Charging of the battery releases positive sodium ions from the sodium polysulfides, sending them back through the electrolyte to recombine as elemental sodium. To keep the electrodes in the molten state, the battery is kept at a temperature of about 570F. NaS modules also include an electric heater to keep the reactants molten, and thermal insulation to reduce heat losses.
NGK’s commercial-scale NaS battery production facility—which opened in April 2003 in Komaki, Japan—has an initial capacity of 65 MW/yr that can be expanded to 200 MW/yr. Modules are currently available with ratings of 50 kW/360 kWh or 430 kWh for peak-shaving (PS) applications and 50 kW/360 kWh or up to 250 kW for short-term power quality (PQ) applications. Units can be combined to provide up to 20 MW for PS, and up to 100 MW for PQ applications. PS modules are optimized to deliver long discharges with modest voltage drop, while the PQ modules are designed to deliver short pulses of power.
The commercial version of the NaS battery has been demonstrated at about 190 sites totaling more than 270 MW in Japan since April 2004. One system installed at a Japanese semiconductor factory was rated at 1 MW/7 MWh for peak shaving duty and supplied up to 3 MW for 13.5 seconds to improve power quality. The system also proved its ability to lower the factory’s cost of energy by shifting some purchases from on-peak to off-peak hours, and to protect important loads from short outages. Another of its benefits was leveling the local utility’s demand.
Today, there are a total of 9 MW of NaS batteries installed in the U.S. In one U.S. demo of the NaS battery, a system was installed in September 2002 at an American Electric Power Co. office building in Gahanna, Ohio. It can simultaneously provide 500 kW for up to 30 seconds for PQ needs and 158 kWh at up to 100 kW for PS. Alternatively, the system can provide 300 kW for 30 seconds for PQ and 720 kWh at up to 100 kW for PS. The largest installation is 34 MW, 245 MWh unit for stabilizing a 51 MW wind farm in northern Japan.
Compared to conventional lead-acid batteries, NaS batteries offer three to five times the energy density and more and deeper discharge cycles. The units are easily sited indoors or out since they are clean, quiet, and vibration-free and their operation is insensitive to ambient temperature. Their major downside is their capital cost; where lead-acid batteries run about $400 to $900/kW, the current NaS modules—which are nominally rated at 50 kW each—are priced at $1,800/kW.
Flow Batteries: Pros and Cons
Flow batteries (FBs) are a relatively new class of electrochemical device. They can store large amounts of electrical energy (from tens of kWh to tens of MWh) and deliver it either slowly over several hours, or within milliseconds or minutes as high-power pulses. The versatility of FBs suits them for a wide range of applications, from energy management to power quality and reliability.
There are three different types of flow batteries, and they use different chemicals to store energy. Two, which use zinc-bromide and vanadium electrolytes, are currently in the early stages of commercialization. Until recently, the third type—the Regenesys battery, which uses sodium-bromide and sodium-polysulfide electrolytes—seemed to have a bright future. A few years ago the German utility giant RWE, Regenesys Technology Ltd.’s corporate parent, abruptly pulled the plug on the R&D effort and a large project with the Air Force was cancelled. Later, RWE sold its intellectual property rights to the technology to Vancouver-based VRB Power Systems Inc. that has since gone bankrupt, so the future of the Regenesys battery is murky at best. The Regenesys Technology website finds their thermal storage interests are focused on integration with solar thermal systems
Flow batteries will have—at least initially—higher capital costs than lead-acid batteries (typically, $350/kWh vs. $200/kWh for a load-shifting application, excluding power conditioning and balance of plant costs), but they also have a much longer cycle life. Accordingly, zinc-bromide and vanadium batteries may be economically attractive to certain users for certain applications. And their cost is certain to fall as production ramps up to commercial levels within the next few years and the economies of scale kick in.
The big operational advantage of flow batteries, which use reversible reduction-oxidation (redox) reactions, is that they allow either power or energy output to be optimized in real time. In a conventional battery, both electrodes are immersed in the same ion-conductive electrolytic solution. During discharge, oxidation at the battery’s negative electrode (anode) liberates electrons. The electrons then leave the anode, flow through the external circuit to which the battery is connected and perform useful work, and are returned to the positive electrode (cathode). At the cathode, reduction recombines the electrons with positive ions in the electrolyte.
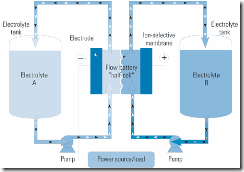
The process is similar in the flow battery, except that each electrode is immersed in a different electrolyte, and the two electrolytes are separated by an ion-exchange membrane. To discharge electricity, the two electrolyte solutions are pumped from separate tanks into half-cells along either side of the membrane (Figure 2). To store electricity, current is put back into the system to return the electrolytes to their original chemical states, recharging the battery.
2. Representative flow battery design. Flow batteries store electrical energy chemically in tanks separated from the electrochemical cells. Each tank contains a different electrolyte solution. Electrolytes are pumped through half-cells, exchanging ions across selective membranes during charging and discharging, producing or absorbing electrons. Source: Platts; adapted from Regenesys Technologies Ltd
NiCad Batteries: Less Costly than They Seem
Nickel-cadmium (NiCd) batteries have powered portable electronic devices for decades. However, their relatively high capital cost has prevented their widespread use in large stationary applications. Nevertheless, there are certain utility market niches in which NiCads can compete on a lifecycle-cost basis with conventional lead-acid batteries because the former can work in extreme temperatures and deliver lots of current over a short period.
Indeed, a phenomenon related to discharge speed is what prompted the Golden Valley Electric Association to choose nickel-cadmium units to power its Battery Energy Storage System (BESS). Commissioned in late 2003, the GVEA system (Figure 3) is the biggest of its kind in the world. With the internal NiCad units connected in series to create a 5,000-VDC battery, BESS is capable of providing 27 MW of AC current for 15 minutes, or up to 46 MW for long enough to start up a backup generator to replace one that fails. GVEA expects about 30 of these events per year in Alaska’s unforgiving climate.
GVEA chose nickel-cadmium batteries to power BESS (which happens to be in a controlled-temperature facility) because their capacity degrades slowly and linearly when they are discharged rapidly.
3. At the heart of the Golden Valley Electric Association’s Battery Energy Storage System are racks holding 13,760 nickel-cadmium modules. Connected in series, the modules function as a 5,000-VDC battery. Courtesy: Golden Valley Electric Association
Perhaps Vanadium Redox
The two major suppliers of vanadium redox batteries are Tokyo-based Sumitomo Electric Industries Ltd. (SEI) and VRB Power Systems Inc. (VPS). Both companies rely on intellectual property held by Sydney-based Pinnacle VRB, Ltd., in which VPS owns a controlling share. Prudent Energy Inc purchased the assets of VPS in January 2009 after VPS declared bankruptcy and now markets the products. In addition, a third company—Reliable Power, Inc.—was authorized by Sumitomo to sell vanadium redox batteries in the U.S. although a recent check found no web site for the company.
SEI has been developing vanadium redox batteries since 1985 and manufacturing and marketing them since 2001. SEI markets megawatt-scale systems directly in Japan, where 11 projects representing more 3.7 MW and 13.8 MWh are in operation.
To date, VPS (now Prudent Energy Inc) has installed eight systems, according to information on their website. One is a 200-kW/1,000-kWh battery connected to a hybrid wind/diesel-powered microgrid on King Island off the south coast of Australia. The other is a 350-kVA/2.4-MWh system (Figure 4) that provides peak shaving and voltage support for a 200-mile-long, 25-kV feeder on the PacifiCorp grid near Moab, Utah; it began operation in March 2004.
4. On the job. VRB Power Systems dedicated this 350-kVA/2.4-MWh vanadium redox battery near Moab, Utah, in March 2004. It provides peak shaving and voltage support on a 200-mile-long PacifiCorp feeder. Courtesy: VRB Power Systems Inc.
Consider Zinc-Bromide
Currently, ZBB Energy Corp. is the only company working to commercialize the zinc-bromide flow battery. The firm has its headquarters and a new production facility in Menominee Falls, Wis., but its research, development, and marketing efforts are based in Perth, Australia. Greg Nelson, the company’s chief operating officer, characterizes the company’s efforts as being in the small-scale demonstration phase.
To date, ZBB has largely focused on demonstrating the technology in the utility market. The company has built energy storage systems whose outputs range from 50 to 500 kWh for PG&E, Melbourne-based United Energy Ltd., Detroit Edison Co., Osaka-based Daihen Corp., and Sandia National Laboratories.
A commercially available 500-kWh grid-interactive storage system is the company’s basic building-block product. One system should satisfy most industrial energy storage needs, and several can be combined into multi-megawatt-hour sizes for utility applications (Figure 5). For example, four of the trailer-mounted systems are scheduled to be installed this year to provide 2 MW of peak-shaving support to a stressed PG&E substation.
5. One-stop shopping for moving and storage. This turnkey zinc-bromide flow battery from ZBB Energy Corp. can supply 250 kW for up to two hours. Delivered inside a standard shipping container, it is trailer-mounted, making it relocatable. Courtesy: ZBB Energy Corp.
Mobile Batteries
Today, frequency regulation is an ancillary service bought by the hour, the day prior to utilization, and dispensed on an as-needed basis by dispatch communiqués and provided by the ancillary service provider on a 15-minute basis. New battery technologies can also mitigate those electricity supply challenges.
Altair Nanotechnologies (Altairnano), a provider of energy storage systems, has developed a lithium-titanate battery system to mitigate some of these ancillary services difficulties. The power storage system provides frequency regulation on a 1-second dispatch basis, as needed.
AES, one of the world’s largest power companies, understood the game-changing possibilities of Altairnano’s technology for mitigating major frequency regulation problems. With AES, Altairnana developed a 2-MW, 500-kWh system with the capability of producing 1,000 amps at 1,000 volts that was designed and built to fit inside two 53-foot trailers. The Altairnano battery system was recently pilot-tested at AES’s Indianapolis Power & Light location. KEMA served as the test contractor, providing independent third-party analysis.
The lithium-titanate battery system exhibits three times the power capabilities of existing batteries and can be described as the combination of a battery and a supercapacitor. This means power can be extracted from as well as inserted into the battery. Altairnano’s lithium-titanate technology is unique because it lacks a solid electrolyte interface (SEI), as shown in Figure 6.
6. A comparison configuration of a typical lithium-ion battery with a new lithium-titanate battery. Source: Altair Nanotechnologies
The SEI is a “film” on the anode that is an internal resistor that limits power output and generates heat in a standard lithium-ion battery. Therefore, the lack of an SEI allows the lithium-titanate battery to work efficiently in extreme temperatures and eliminates thermal runaway risk. This battery’s operating temperature range also is wider than that of other technologies: from –40C to 55C (–40F to 131F). This capability eliminates the need for supplemental heating when the battery is used in low-temperature environments.
Inside the AES 53-foot trailer (Figure 7) reside numerous Altairnano Super Modules installed in racks (Figure 8). The batteries are air-cooled (the black circles in Figure 9 are the fans) in order to mitigate I2 R heating in the modules. An air-conditioning system for the trailer keeps the temperature below 55C (131F).
7. The Altairnano/AES 1-MW system can produce 250 kWh and is contained within a single 53-foot trailer. Courtesy: Altair Nanotechnologies
8. The batteries are arranged in racks within the trailer along with air-cooled fans, air conditioning, and various computer monitoring systems and controls. Courtesy: Altair Nanotechnologies
Various computer systems monitor and control all the Super Modules and associated systems. A battery management system monitors battery cell temperatures, balances the cell voltages as needed, and keeps track of the battery charge state. A programmable logic controller interfaces with three single-phase Parker Hannifin SSD inverters, one for each phase of the three-phase system. The Parker Hannifin SSD power inverters were coupled to isolation transformers and fed into a step-up transformer with the battery side running three-phase 480 V and the grid side operating at 13.8 kV. A supervisory control system monitors and interfaces with the step-up transformer sending and receiving power from the grid.
Independent Test Results
The AES pilot test at Indianapolis Power and Light was considered by all a significant success in the application of power storage devices for grid application. According to KEMA, the prototype units in their current state are suitable for use in future market pilot activities designed to help better define the application requirements and demonstrate the potential of this technology.
KEMA’s report noted that, “A key performance finding was the maximum unit storage capacity for each of the two one megawatt systems was approximately 300 amp-hours with a capacity of delivering 250 kWh at a rated output of 1,000 kW for 15 minutes. Each unit was able to dispatch at any power level between 1-MW discharge to 1-MW charge within one second. Due to the battery and inverter technology used, response actually occurs within cycles. The round trip efficiencies are on the high end of various options. It is important to note that even with a 90% round trip efficiency, a 250 kWh system requires the replacement of 25 kWh when the total discharge is considered. Other systems that have a 60 to 70% round trip efficiency will not provide the economics necessary when the losses are included in the calculations. Also, the Altairnano system has over a 7-year life when the CAISO standard dispatch model is applied. The cycle life of other electrochemical systems cannot approach this lifetime.”
Next Generation: Air-Fueled Battery
Researchers around the world, meanwhile, are reporting breakthroughs on existing and novel technologies. The University of St. Andrews in the UK, collaborating with colleges from Strathclyde and Newcastle, in May claimed to have designed a new type of air-fueled battery that can provide up to 10 times the energy storage when compared with designs currently available.
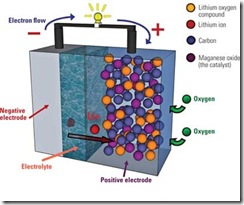
The STAIR (St. Andrews Air) cell capacity is based on rechargeable lithium batteries, which are currently composed of a graphite negative electrode, an organic electrolyte, and lithium cobalt oxide as the positive electrode. Instead of lithium from the layered intercalation compound (lithium cobalt oxide), the STAIR uses a porous carbon electrode. The oxygen, which will be drawn in through a surface of the battery exposed to air, reacts within the pores of the carbon to discharge the battery. The university has discovered in the course of its four-year study that the carbon component’s interaction with air can be repeated, creating a cycle of charge and discharge (Figure 9).
9. Researchers from the University of St. Andrews in the UK have designed an air-fueled battery that they claim could last 10 times longer than designs currently available. As the diagram of the lithium-air STAIR (St. Andrews Air) cell shows here, oxygen is drawn from the air and reacts within the porous carbon to release the electrical charge. Courtesy: University of St. Andrews
Initial results from the project found a capacity to weight ratio of 1,000 milliamp-hours per gram of carbon (mAh/g), while recent work has obtained results of up to 4,000 mAh/g, the researchers said. The researchers expect that the battery is about five years away from commercial availability, however.
In May last year, a Canadian research team at the University of Waterloo reported it had laid the groundwork for a lithium-sulfur battery that could store and deliver more than three times the power of conventional lithium ion batteries. As reported in the online issue of Nature Materials, the team overcame the challenge of keeping the electrically active sulfur in contact with a conductor, such as carbon. The team chose — at a nanoscale level — a member of a highly structured and porous carbon family called mesoporous carbon.
Filling the tiny voids then proved simple: Sulfur was heated and melted. Once it came into contact with the carbon, it was drawn or imbibed into the channels by capillary forces, where it solidified and shrunk to form sulfur nanofibers. Scanning electron microscope sections revealed that all the spaces were uniformly filled with sulfur, exposing an enormous surface area of the active element to carbon and driving the exceptional test results of the new battery. The research team continues to study the material to work out remaining challenges and refine the cathode’s architecture and performance.
Portions of this article were previously published in POWER magazine and POWERnews. Sr. Writer Sonal Patel also contributed to this article.






Although it’s good to see this kind of innovative panoply, one should note that, while the birth announcements of such devices appear abundantly , their consequent obituaries are rarely observed.
One should also abjure the prospect of building expansive “renewables” all the hell over the place” in the hope that, one day, somehow, this storage technology will make them all work properly. Such “build it and the enabling technology will come” exuberance should be anathema to informed practice.
Robert:
Thank you for an informative article.
The waxing on about batteries of the future can’t help but remind me of the promises made about nuclear fussion: on paper things seem great. In reality it is an entirely different matter.
And, of course, the cost projections of industry lobbyists and agenda promoters can’t be considered credible.
I also can’t help but think of the analogy of an engineering expert who I heard talk about batteries for such sources as industrial wind power. He said: “another word for a battery for such an application, is bomb.”
Vanadium flow batteries will soon have another positive on their side – a 200 Mt vanadium deposit has just been discovered in Africa that is sediment-hosted This means a cost o produce of less than $2.00/lb. That’s enough vanadium to supply the world batteries for decades. The company that owns this deposit is Energizer Resources (URST.OB) http://www.energizerresources.com Their Green Giant vanadium deposit will offer sureness f supply and cost to future vanadium redox battery manufacturers. Given this and the comprehensive info in your article my money is on vanadium.
Check these links about vanadium flow batteries…
The Vanadium Battery: The Ultimate Energy Storage Solution – http://tiny.cc/UNo8J
Vanadium – The element that could change the world: http://tiny.cc/gdmbO
I got as far as “MW/year” and then I stopped and pondered. Then I concluded to stop reading. Units of power per time period kind of ground me to a halt. I could have hung with MW years, but not MW per year. I want to know more about batteries someday.
Dear Sir,
We are interested to know who will be the authorized dealers throughout the world to market STAIR batteries. Who will be the authorised dealer/agent for Bangladesh ?
What will be price ranges according to power?
We think it is the last blessing to the mankind.
Awaiting your kindest reply asap.
Thanking you all.
[…] supply and demand must match continuously. Unlike the water supply, large amounts of electricity can’t be stored, despite century old quests, led by Edison, to invent such battery storage systems. AWEA’s […]
Your title says that there is no silver bullet battery for renewables, but where are the conclusions in your article?
I am interested to learn how I can implement solar+wind energy + storage of this energy as an individual, not as a company.
Where can I find a “how to” using existing techniques?
Any info on this subject would be much, much appreciated.